32. 80 g of h2 is reacted with 80 g of o2 to form water. find out the mass of water obtained.which substance is the limiting reagent.
4.6 (166) In stock
32. 80 g of h2 is reacted with 80 g of o2 to form water. find out the mass of water obtained.which substance is the limiting reagent.
32- 80 g of h2 is reacted with 80 g of o2 to form water- find out the mass of water obtained-which substance is the limiting reagent
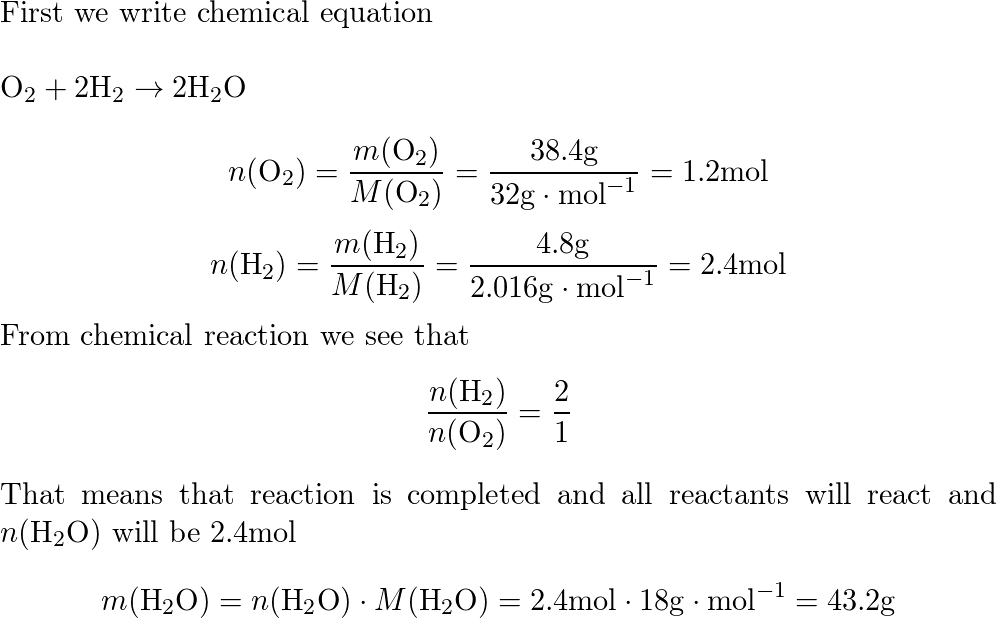
Hydrogen and oxygen react chemically to form water. How much

41. 8g H2 and 32 g 0, is allowed to react to form water then which of the following statement is correct (1) O, is limiting reagent (2) O, is reagent in
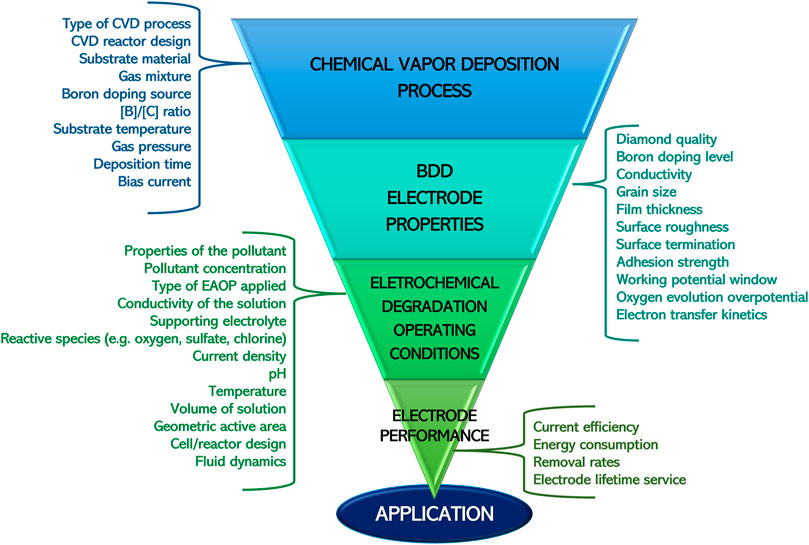
Frontiers In-house vs. commercial boron-doped diamond electrodes for electrochemical degradation of water pollutants: A critical review
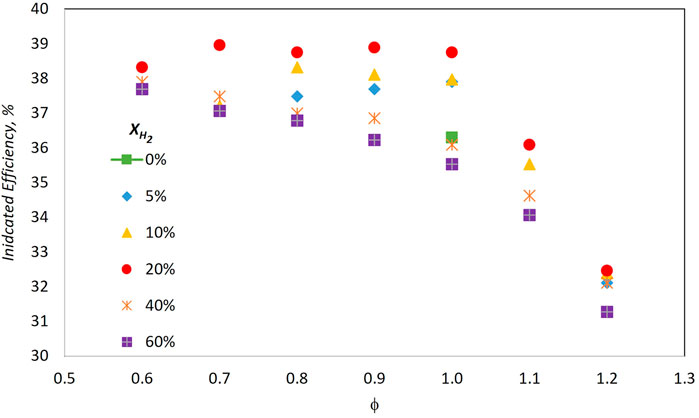
Frontiers Ammonia as Green Fuel in Internal Combustion Engines: State-of-the-Art and Future Perspectives

80 g of H_2 is reacted with 80 g of O_2 to form water. Find out the mass of water obtained . Which substance is the limiting reagent ?

Hydrogen and oxygen combine in the ratio of 1:8 by mass to form water

Chapter 11

Solved Butane, C4H10, reacts with oxygen, O2, to form water

Spatiotemporal Decoupling of Water Electrolysis for Dual-Use Grid Energy Storage and Hydrogen Generation - ScienceDirect

iii. Mass of mathrm{CO}_{2} remaining =319 mathrm{g} Q.88. 6 mathrm{g} of mathrm{H}_{2} reacts with 32 mathrm{g} of mathrm{O}_{2} to yield water. Which is the limiting reactant? Find the mass of water produced

Review of the Decomposition of Ammonia to Generate Hydrogen
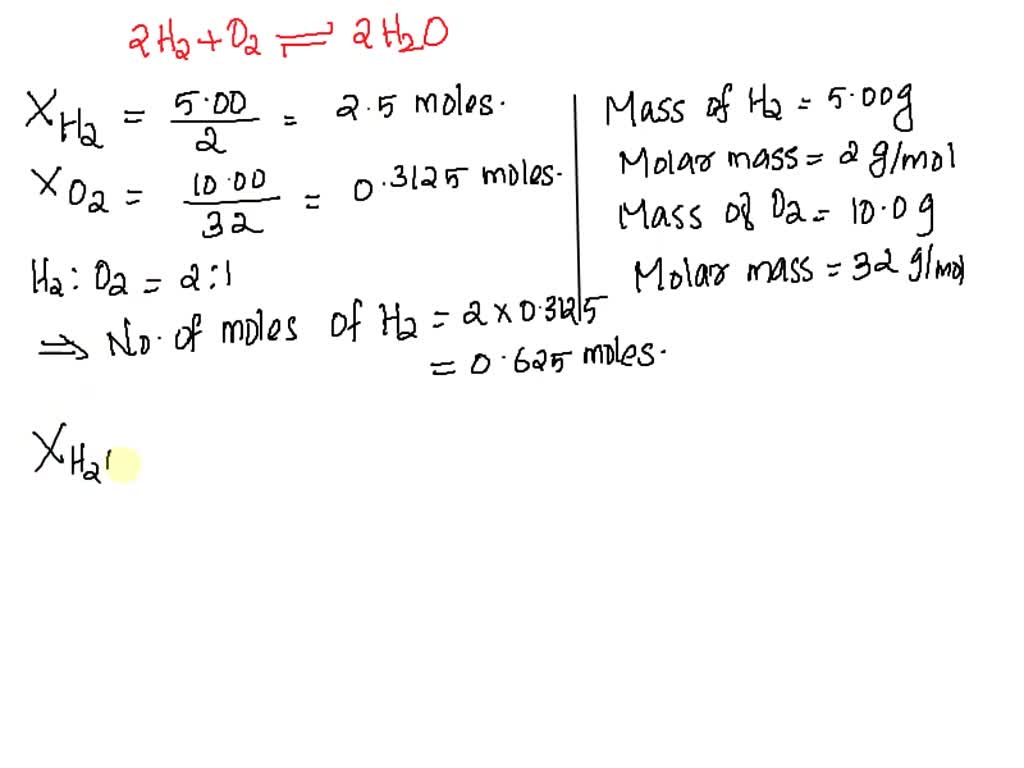
SOLVED: Which is the limiting reactant when 5.00 g of H2 and 10.0 g of O2 react and form water? How many grams of water are formed and what mass of excess

Interface, Vol. 32, No. 2, Summer 2023 by The Electrochemical Society - Issuu
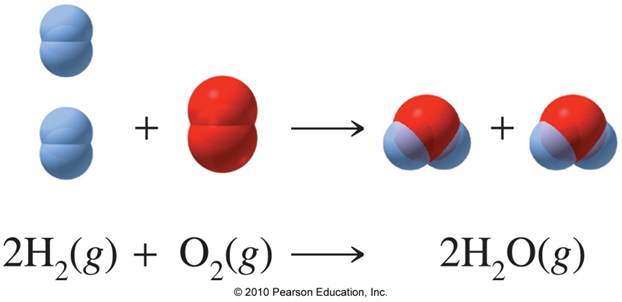
How many grams of water can be produced the combination of 8 grams of oxygen and 8 grams of hydrogen?
BD Ultra-Fine™ Micro 6mm x 32 G pen needle
Chocolate Trento Branco Dark c/16 un x 32 g PECCIN - Distribuidora Girotto, Doces e Embalagens
Tablet Vasoun Kids 7 Polegadas Tablet para Crianças Android 11 Tablet 2 Gb Ram 32 Gb
Buy Amul Butter Cookies 32 g (Pack) Online at Best Prices in India - JioMart.




